
In 1896, Birkeland offered a radically new perspective on theĪuroral problem, suggesting that cathode rays from the Sun excited theĮmissions (Egeland and Burke, 2005). Particle orbits that simulated Birkeland's Terrella experiments (Egeland and Burke, 2012). Later he came to admire Størmer's calculations of auroral Particles were radioactivity and cathode rays.Īuroral scientists Kristian Birkeland and Carl Størmer (1874–1957) At this time, the only known sources of energetic His 1911 paper in Nature was “The Radiation Producing Aurora Borealis”. (alpha rays) bombarding the upper atmosphere (Vegard, 1911). His hypothesis that the aurora could be produced by energetic helium ions During this time, he was assembling arguments in support of He wished to studyĮmissions generated by cathode and anode rays for comparison with auroral Joseph Jon Thomson and Wilhelm Wien, respectively. In 19, Vegard conducted advanced spectroscopic studies atĬambridge and Würzburg universities under the direction of Professors This review concentrates on hydrogen emissionsĮxcited by energetic protons bombarding the auroral ionosphere. 
States (Omholt, 1971 Vallance Jones, 1974). Mechanisms other than transitions between O( 1S) and O( 1D) atomic Period 1950–2000, numerous papers discussed different indirect excitation Line was not reproducible in a laboratory until 1924 (McLennan and Shrum, 1925). Vegard alsoĬontributed to the development of competing excitation models proposed toĮxplain the generation of the auroral green line emissions. He is widely known for identifying several auroralĮmissions associated with atmospheric nitrogen (Vegard, 1956). Specify the auroral green line's accurate to the wavelength 557.7☐.1 nm (Vegard, 1923 Babcock, 1923). Aroundġ920 he devised a double-prism spectrograph with resolution sufficient to He soonĭeveloped a high-quality scientific laboratory at the university. Of physics at the University of Kristiania (now Oslo) in 1918. Subsequent to Birkeland's death, Vegard was appointed professor Narrative, began to work as an assistant to professor Kristian Birkeland In 1906 Lars Vegard (1880–1963), a central character of the present Officially as being equal to 10 −10 m (Franke, 1969). He introduced the wavelength unit, 1 Å = 10 - 8 cm, which appeared Ångstrøm (1868) used a theodolite with a transmission grating whenĭiscovering the auroral green line at 5578 angstrom ( Å). Pioneering spectroscopic work by Anders Ångstrøm (1814–1874) in theġ850s marks the beginning of quantitative spectral studies as applied to the In solar wind dynamic pressure or the polarity of the north–south component Imagers showed that dayside hydrogen emissions appear in response to changes Prevail, allowing them to precipitate through filled loss cones. Of regions in the magnetosphere, conjugate to nightside proton aurora, whereĬonditions for breaking the first adiabatic invariants of kiloelectron volt protons Space-age data from satellite-borne sensors made two significantĬontributions: (1) energetic particle detectors demonstrated the existence Hydrogen radiation came from altitudes between 105 and 120 km. Sounding rocket data showed that most of the Generated hydrogen atoms were left in excited states and emitted the They undergo charge-exchange events with atmospheric neutrals. Ultimately of solar origin, precipitate into the topside ionosphere, where These observations in turn, suggested that protons, Separations between the locations of electron- and proton-generated aurorae Indicated that emitting particles had energies well into the kiloelectron volt range,Ĭorresponding to velocities >1000 km s −1. Α (121.6 nm) hydrogen emissions, were also detected.ĭoppler blue shifts in hydrogen emissions, established in the 1940s, Satellite observations, the Balmer lines H δ and H ε at 410.13 and 396.97 nm, respectively, as well as extreme ultraviolet (EUV) Lyman Identified in ground-based measurements prior to the space age. 
Lines of the Balmer series, including H γ at 410 nm, were Spectroscopists Vegard, Gartlein and Meinel investigated otherĬharacteristics of the auroral hydrogen emissions. 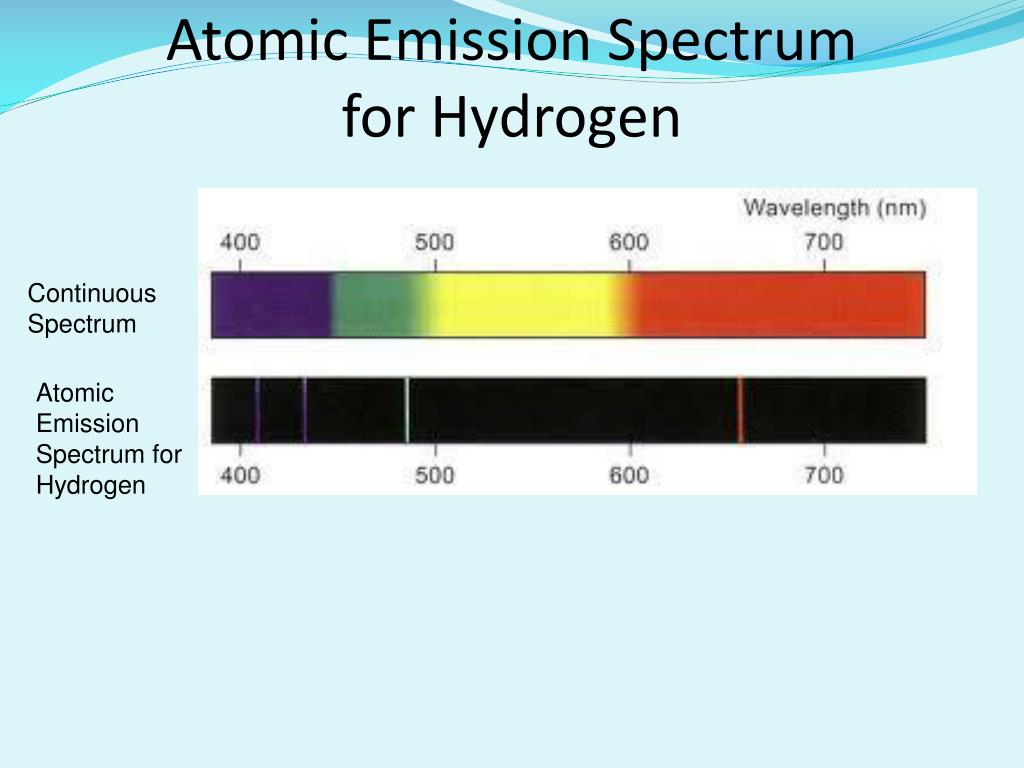
With more advanced instrumentation after World War II, auroral Positive bands of N 2 + nearly covered the H α emissions. Structure, located equatorward of the auroral zone. Lars Vegard discovered the H α (656.3 nm) and H β (486.1 nm) spectral lines of Balmer series emissions, emanating from a diffuse Ranks among the first steps toward developing this tool. Of discrete lines whose wavelengths follow a simple mathematical pattern, which In 1885,īalmer discovered that the visible hydrogen spectrum consists of a series Auroral spectroscopy provided the first tool for remotely sensing theĬompositions and dynamics of the high-latitude ionosphere.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
